LonelyStoner
Member
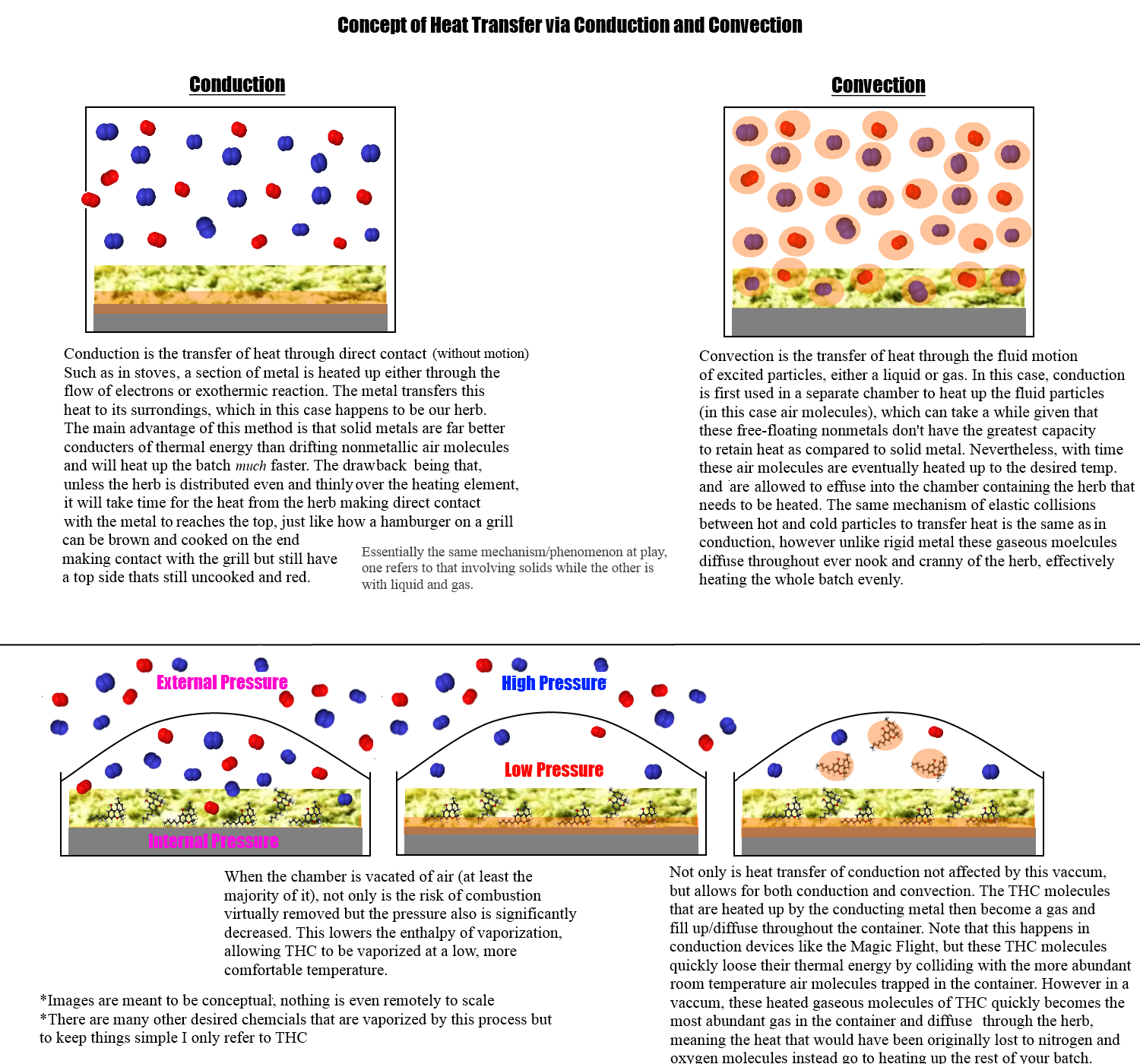
The most prevalent problem I found with vaporizers is accidental combustion. I mean, this seems like a pretty easy problem to fix. All you need is a vaporizer with a chamber that can be vacated of air (otherwise establish a vacuum) before beginning to vaporize. The heating element will still be able to force THC and other substances to undergo the phase change to a vapor, but while lacking any oxygen needed to react with the plant matter to cause combustion. Of course, air would have to be reintroduced at the end to clear out the chamber but still seems like an easy and reliable concept
Are there any vaporizers like this? If not, why? Is there something I'm overlooking...
Are there any vaporizers like this? If not, why? Is there something I'm overlooking...